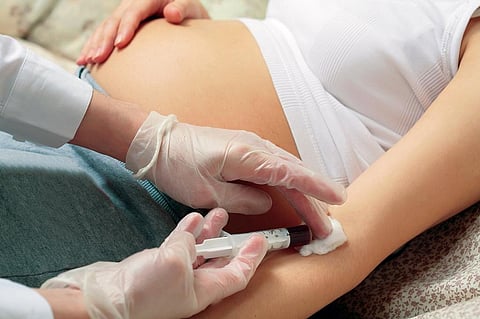

Oxytocin is a hormone that women produce naturally during and after childbirth and is said to strengthen the bond between mother and child. But many women have to be given the hormone after vaginal childbirth to treat blood haemorrhage. Gynecologists often prescribe the hormone to strengthen contractions during childbirth and control bleeding after childbirth. In fact, the drug is part of India’s National List of Essential Medicines.
While the Union Ministry of Health and Family Welfare had earlier decided to restrict the use of this drug, it later allowed private retailers to sell the drug fearing shortage. Earlier, after September 1, 2018, only one public sector manufacturer, the Karnataka Antibiotics & Pharmaceuticals Limited (KAPL), was allowed the licence to produce this hormone and it could only sell this through registered hospitals under the Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP) and the Affordable Medicines and Reliable Implants for Treatment (AMRIT). But, after the recent turnaround, the hormone can be sold by private retail chemists but with strict regulations.
The new rules, issued on Tuesday (August 21), state that oxytocin be moved from schedule H to schedule H1 from September 1. This means that while the buyer earlier just needed a prescription from a medical practitioner to get the medicine, the new rules requires the chemist to maintain a record of name and address of the prescriber and the patient as well as the drug’s name with the quantity sold.
Reason for earlier restriction
In October 2014, the Union Minister of Women and Child Development Maneka Gandhi wrote to the health ministry to flag the issue of “loss of livestock in the country” due to the illegal use of oxytocin. It is no secret that the dairy industry uses the drug to increase milk production in cattle. On March 15, 2016, the Himachal Pradesh High Court asked if restricting the manufacture of oxytocin only in public sector companies was feasible. The Drugs Technical Advisory Board, a statutory body constituted under the Drugs and Cosmetics Act, 1940, recommended that formulations for human use can be restricted to be supplied only to registered hospitals in the public and the private sector to prevent its misuse. At present, oxytocin used by the livestock industry is produced in unlicensed facilities.
But health activists label the move as an effort towards gauraksha, disregarding woman’s health. The president of the Federation of Obstetric and Gynaecological Societies of India (FOGSI), Jaideep Malhotra, says that the maternal mortality ratio (MMR) is likely to increase due to this illogical decision. She points out this will create a problem for small clinics that do just five to 10 deliveries a month as they would now have to set up cold storages to preserve them. “About 70 per cent of deliveries are done in private clinics, but they have not been kept in mind while taking the decision. What is the backup plan if a natural disaster hits KAPL and production has to be stopped?” she asks.
Activists point out other issues with KAPL. For one, it does not have any experience in making and supplying the drug as they began production for the first time only on July 2 this year. It is estimated that around 60 million ampoules of oxytocin would be needed every year. The price of the drug being quoted by KAPL is higher than what states are paying now and thus will put states under financial burden. KAPL doesn’t even have expertise in cold chain which is essential to prevent degeneration of oxytocin. “Our experience with paediatric aids medicines show that the government’s cold chain is plagued with supply chain issues,” says Leena Menghany of Médecins Sans Frontières (MSF), a non-profit. FOGSI has sent its concerns to the health ministry. Activists too have filed a public interest petition through the All India Drug Action Network (AIDAN) in the Delhi High Court, asking for the order to be quashed. On August 16, 2018, the court asked the government to respond before August 24.
As of now, there is no research to prove that misuse of oxytocin by the dairy industry is impacting human health. A study by the National Institute of Nutrition, Hyderabad, and another by the National Dairy Research Institute, Karnal, says that the presence of oxytocin in milk for human consumption was undetectable due to degradation in the intestine. At the same time, there are anecdotal reports about the ill-effects of oxytocin on cattle. It is said that oxytocin leads to infertility in cattle. It has also been linked to mastitis, a painful inflammation of the udder. Health activists say that concerns about animal health should be dealt without affecting the availability, accessibility and affordability of oxytocin for women. The dairy industry is misusing the drug, not the doctors, says Malhotra.
WHO, which has been quiet on the issue so far, recently pointed out that the benchmark for cervical dilation rate at 1 cm/hr during the active first stage of labour is unrealistic for some women and should not be used as an indicator for the use of oxytocin to hasten labour. This would reduce excess and irrational use of oxytocin. To take care of haemorrhage cases, who along with Ferring Pharmaceuticals and MSD for Mothers have tested a new drug carbetocin and have found it to be as effective as oxytocin in controlling haemorrhage. An additional advantage is that the drug is heat stable, unlike oxytocin. Researchers say the drug would help reduce MMR due to haemorrhage in regions that don’t have cold storage facilities. This is where 99 per cent of maternal deaths due to haemorrhage currently occur. However, it will take time before carbetocin reaches the markets, as Ferring is seeking registrations for its manufacture. Other than oxytocin, the choices for women include Methergine which too requires cold storage and Misoprostol, an oral drug which has side-effects.
It is difficult to understand what the government’s plan is. On August 10, it released a draft notification that shifted oxytocin from Schedule H to Schedule H1 of the Drugs and Cosmetics Rules, 1945. This would mean more documentation would be needed at the retailer level suggesting the government could be considering sales through retail chemists. It must be noted that the government has not taken similar action in case of antibiotics which are being misused by the poultry industry. “The decision seems hasty for many reasons,” says Malini Aisola of AIDAN.
(This article will be published in the 1-15 September 2018 edition of Down To Earth’s print edition)