
The more we learn about the science behind COVID-19, the more we are beginning to understand the vital role a single molecule in our bodies plays in how we contract the disease.
That molecule, Angiotensin Converting Enzyme 2, or ACE2, essentially acts as a port of entry that allows the coronavirus to invade our cells and replicate. It occurs in our lungs, but also in our heart, intestines, blood vessels and muscles.
And it may be behind the vastly different death rates we are seeing between men and women.
What is ACE2?
ACE2 is an enzyme molecule that connects the inside of our cells to the outside via the cell membrane.
In normal physiology, another enzyme called ACE alters a chemical, Angiotensin I, and converts it into Angiotensin II, which causes blood vessels to constrict. The tightening of the blood vessels leads to an increase in blood pressure.
That’s when the ACE2 molecule comes in: to counteract the effects of ACE, causing blood vessels to dilate and lowering blood pressure.
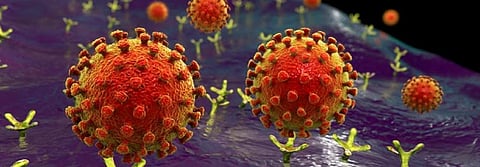
You may have seen illustrations of the virus that show distinct spikes around the surface of the virus, which form part of the “crown” or “corona” that gives the virus its name. These spikes are called S1 proteins, and they are what binds to the ACE2 molecule on our cells.
The virus is then able to invade the cell by a process called endocytosis — where the cell membrane engulfs the virus and internalises it within a bubble called an endosome.
Once inside the cell, the virus interacts with the host cells’ genetic machinery, taking advantage of the existing structure to replicate extensively.
SARS-CoV-2, the virus behind COVID-19, has a high binding capacity for ACE2 — between 10 and 20 times more that of the original SARS virus. This means it is much easier for SARS-CoV-2 to get into human cells compared to the original coronavirus, making it more infectious overall.
ACE2 and COVID-19
But there is still conflicting evidence on the precise role ACE2 plays in coronavirus infections.
In some cases, it can actually be of benefit: ACE2 has been shown to reduce injury to the lung tissue in cases of the original SARS virus in mice by doing its job and causing blood vessels to dilate.
In another mouse study, however, the binding of the SARS spike protein to ACE2 was shown to contribute to lung damage.
When it comes to the current coronavirus, early studies have shown that the introduction of a human-made form of ACE2 to human cells can block the early stages of infection by binding the spike protein, preventing it from entering the cells. ACE2 thus acts both as an entry port to cells but also as a mechanism to protect the lung from injury.
ACE2 and the male death toll
It’s well established that COVID-19 affects men and women differently. Out of a representative sample of 1,099 patients in China, 58 per cent were men and 42 per cent were women. Data from China has also shown that men die of COVID-19 at a rate two-and-a-half times that of women.
Similar figures have been observed in the United States and 60 per cent of deaths in Europe have been men.
We don’t yet fully understand why men die of COVID-19 in higher numbers than women, but it’s possible ACE2 plays a role.
A large study of two independent populations of heart failure patients was recently published in which ACE2 concentrations were found to be significantly higher in men than in women. This could explain why men may be more at risk than women of COVID-19 infection and of dying from the disease.
ACE2 and other conditions
The other important factor in the outcome of patients is the presence of underlying health problems.
Recently, ACE2 has been identified in different cells of the heart. There are a greater number of ACE2 receptors on the surface of cells in the heart muscle in people with established cardiovascular disease compared to those without disease.
This may result in a greater number of virus particles entering the heart cells in COVID-19 patients with established heart diseases.
Given the role ACE2 plays in regulating blood pressure, there are also concerns about how it affects COVID-19 patients with hypertension. Men are more likely to have hypertension than women, especially under the age of 50.
Two particular drugs to reduce hypertension also affect ACE and ACE2. These are Angiotensin Converting Enzyme inhibitors, or ACEi, and angiotensin receptor blockers, known as ARBs. In animal studies, both of these drug types increase the production of the ACE2 enzyme and so may increase the severity of COVID-19 infection.
Small independent studies have examined the effect of these treatments on COVID-19 with conflicting results. However a recent study on the subject has demonstrated that COVID-19 patients with untreated hypertension have a higher risk of death compared to those being treated with ACEi or ARBs.
That’s why many professional societies are advising people with high blood pressure to continue using their medicines during the crisis.
The role that ACE2 plays in COVID-19 is important in our understanding of the disease and could be used as a target for therapy. Drugs could be designed to block the receptor function of ACE2, but also there is promise in using the molecule itself in preventing entry of the virus into cells.
This would protect organs such as the lung, heart, kidney and intestine from extensive damage, and hopefully reduce mortality.
This article is republished from The Conversation under a Creative Commons license. Read the original article.